Does Growing New Neurons Improve Memory?
Post by Rebecca Glisson
The takeaway
Recent studies have shown that humans can grow new neurons (neurogenesis) even as adults. Individuals with Alzheimer’s disease have much lower neurogenesis than normal, while older adults with high cognitive capacity have abnormally high neurogenesis.
What's the science?
It was long thought that adults do not grow new brain cells (neurogenesis), but more recently, we have come to understand that neurogenesis happens throughout our lives. Most studies use rodents as a model for human brains, which doesn’t yield enough insight into how neurogenesis occurs in adult humans. This week in Nature, Disouky and colleagues studied the brains of humans at all ages, with and without Alzheimer’s disease, to better understand the link between neurogenesis and cognitive functioning.
How did they do it?
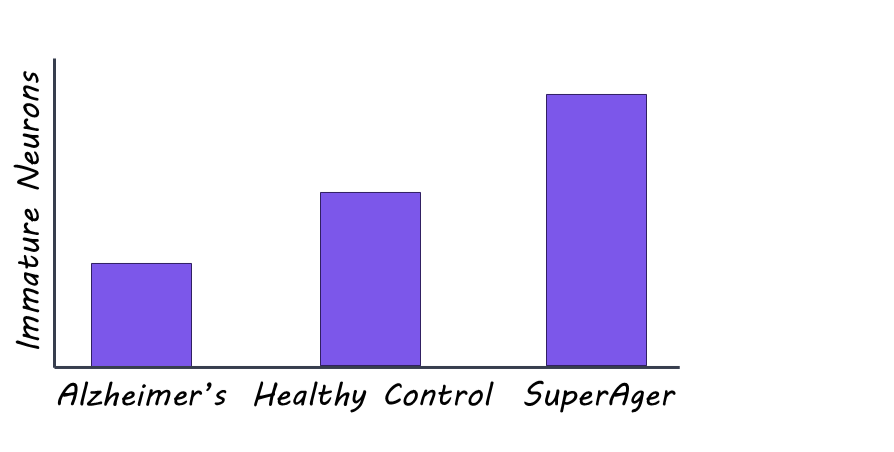
To study neurogenesis in humans, the authors used the brains of deceased donors. The donors were groups of individuals at different ages and cognitive abilities, including young and older adults with normal cognitive ability, individuals who had or were starting to show signs of Alzheimer’s disease, as well as adults they called “SuperAgers”, or individuals who had high memory test scores. The authors identified neurogenesis from the genetic sequence present in cells in the hippocampus, a part of the brain involved in memory, using a method called RNA-seq. Cells that have just grown, immature cells, have a specific genetic sequence that the authors analyzed to determine how much neurogenesis had occurred.
What did they find?
Both younger and older adults with healthy cognitive ability had high levels of neurogenesis in their hippocampi. In contrast, individuals with Alzheimer’s disease or symptoms of it had less neurogenesis and fewer immature neurons. This suggests that the slowing of neurogenesis may impact memory in diseases such as Alzheimer’s. One interesting result was that the “SuperAger” adults had significantly more neurogenesis and more immature neurons than any other group, including the healthy adults, suggesting that neurogenesis may help support healthy cognitive functioning as we age.
What's the impact?
This study is the first to analyze the sequence of new immature neurons in humans and the first to link neurogenesis to cognitive function in individuals of different age groups and cognitive ability. Studies like these can help us better understand mechanisms of memory preservation or memory decline and how to develop better preventative care and treatment for those impacted.