How Do Pain Pathways Drive the Placebo Effect?
Post by Lila Metko
The takeaway
Placebo pain reduction is a phenomenon where prior experience or expectations suppress pain in response to the administration of an inactive treatment. Placebo reduction of pain involves input from multiple cortical regions to the brainstem, which gates brainstem endogenous opioid release, reducing the experience of pain.
What's the science?
Placebo analgesia (reduction of pain) is well known for its ability to complicate experimental procedures. It can also be a useful phenomenon relevant to therapeutic development. For example, if systems involved in placebo analgesia are understood, clinicians may be able to provide treatments that deliberately engage them to provide pain relief. This week in Neuron, Livrizzi and colleagues reverse translate a human placebo conditioning paradigm to mice, and uncover cortex to brainstem connections that gate release of endogenous opioids to downstream pain-modulatory regions.
How did they do it?
The authors used a conditioning paradigm where contextual cues were paired with either morphine + pain stimulus or saline injection (placebo) + pain stimulus. For the morphine conditioning, the idea is that in the absence of morphine, these contextual cues would trigger placebo analgesia in the mice. After conditioning, the placebo test included placing these conditioned mice in similar contexts to the morphine conditioning, but with a saline (placebo) injection to induce placebo analgesia. They measured pain in animals by how long it took them to remove their paw from a pain-inducing apparatus (withdrawal latency). The tools they used to manipulate and record pathways involved in placebo analgesia were chemogenetics to activate pathways and fiber photometry to record opioid signaling. The authors used TRAP2 mice - mice that have genetic modifications that allow for labelling and then selectively analyzing neurons involved in a certain behavior or process. In this case, that behavior was placebo analgesia. They also used an interesting approach called in-vivo drug uncaging, which allowed the release of an opioid receptor antagonist over a specific temporal window, in this case, the placebo analgesia test window.
What did they find?
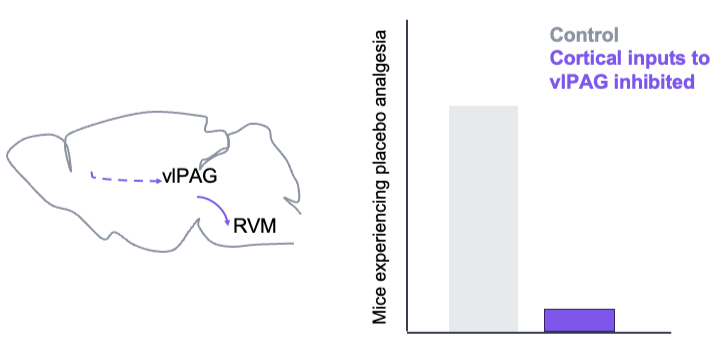
This study found that the vlPAG (ventrolateral periaqueductal grey), a brainstem region with glutamatergic neurons that activate to produce analgesia, is involved in placebo analgesia. They also found that neurons active in the vlPAC during placebo analgesia receive projections from neocortical and insular regions, while neurons in the rostroventral medulla (RVM) received projections from PAG analgesia neurons. After further experiments, they found that placebo analgesia was reduced when the neocortical regions, and not the anterior insular regions, were inhibited. Similar findings occurred when the PAG to RVM connections were inhibited, although both morphine and placebo nociception were altered in this case, not just placebo nociception. They additionally showed that placebo analgesia can be transferred between multiple pain modalities.
What's the impact?
This is the first research study to provide causal evidence of circuits involved in placebo analgesia. Importantly, it moves from correlational human evidence of cortex to brainstem circuits being involved in placebo analgesia to causational data using animal models. Understanding this circuit, especially its role in lasting analgesia after injury, opens up possibilities for future therapeutics.