How the Hippocampus Combines Place and Emotion in Memory
Post by Anastasia Sares
The takeaway
In this study, the authors show that different parts of the hippocampus, a memory formation structure in the brain, work together to bind environmental location and emotional information to form memories of reward or danger.
What's the science?
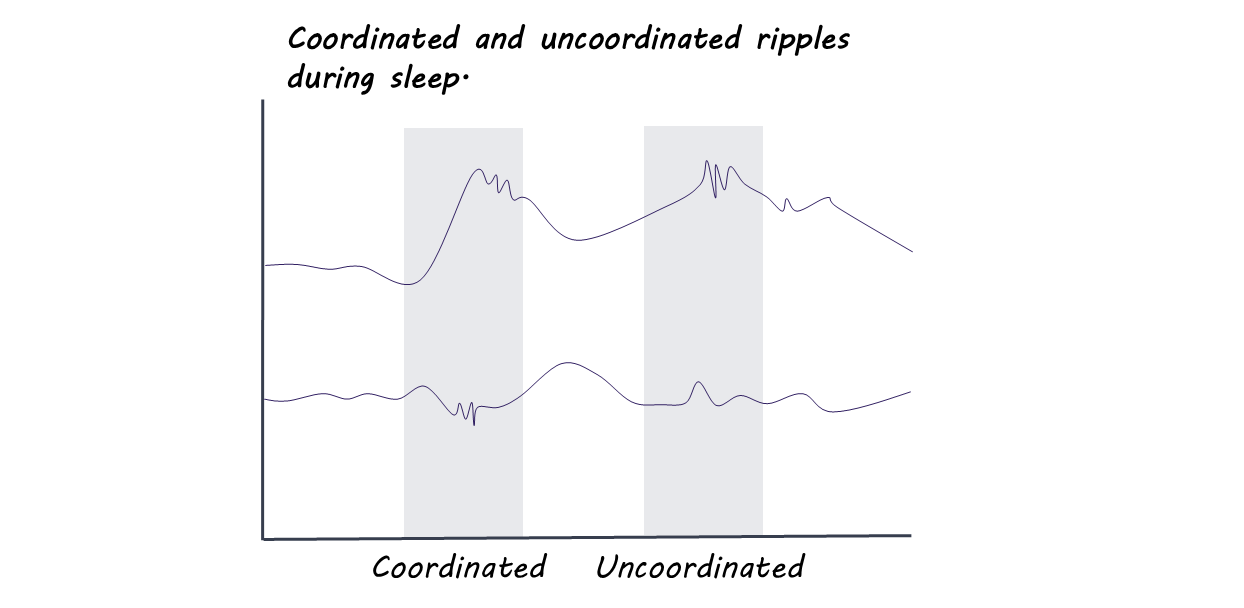
The hippocampus is a brain structure involved in memory formation. When an animal is navigating its environment, cells in the dorsal (“upper”) hippocampus called “place cells” fire when the animal is in different locations. Then, when an animal is sleeping, these cells fire again, in special bursts called “sharp-wave ripples.” This process reactivates other parts of the brain that were involved in the experience, and this is one way scientists think memories are consolidated and strengthened.
However, the hippocampus is not just for remembering where we’ve been. The ventral (“lower”) hippocampus has strong connections to the limbic system, a collection of deep brain structures that helps us process emotion. The limbic system includes the amygdala, which is involved in fear and other strong emotions, and the ventral tegmental area, which is involved in reward and motivation. This week in Nature Neuroscience, Morici and colleagues proposed that the ventral hippocampus is a key linking structure that helps us associate specific emotions with specific environments, driving behavior.
How did they do it?
The authors recorded brain activity from both the dorsal and ventral hippocampus of rats as they completed a navigation task, either to avoid danger (in the form of mild shocks) or to get a reward (in the form of a drink). They recorded this activity both as the animal was learning the task, as well as when the animal was sleeping that night and consolidating their memories from the day.
During learning, both the dorsal and ventral hippocampus were active, but the authors were specifically interested in whether they were working together or not. They used independent component analysis to identify patterns of coherent neural activity, which the authors called “assemblies.” They then tracked these assemblies during learning and during sleep.
What did they find?
During learning, the authors found 446 neural assemblies with coordinated activity. About 62% of these reflected activity mainly in the dorsal hippocampus, 14% reflected activity mainly in the ventral hippocampus, and 24% reflected activity in both regions (which they called “joint assemblies”). These joint assemblies looked very different between the reward and danger conditions, potentially indicating that the hippocampus was changing its internal connectivity depending on the emotional content of the experience. About half of the assemblies were associated with rewarding experiences, and half with dangerous experiences.
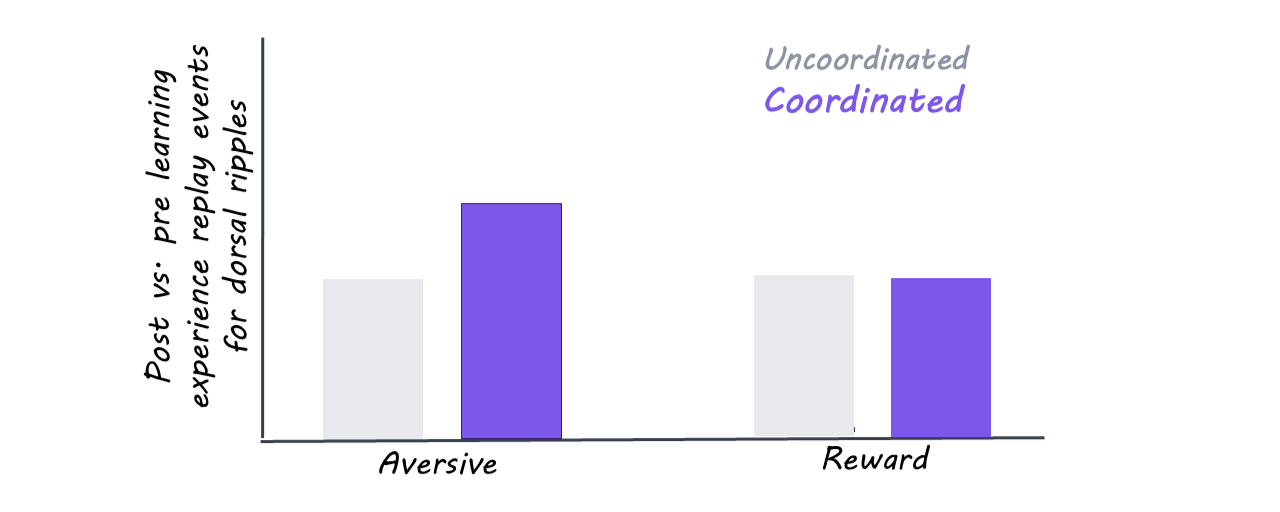
During sleep, the authors continued to track neural activity in both regions. Many of the ripples, those bursts of activity that help reactivate memories, were coordinated between the dorsal and ventral hippocampus. Depending on the experience of the day, different assemblies would be activated: specifically, on the days the animal had been learning from shocks, the cells in the ventral hippocampus responding to danger were more coordinated with place cells in the dorsal hippocampus. Again, this shows that the hippocampus may have different internal connectivity when linking a memory to reward or to danger.
What's the impact?
This work helps us understand the mechanism that links environmental location to emotional memories. It helps us tie what we know about the binding of memories to specific, anatomically connected parts of the brain.