Brain Rhythms Translate Empathy Into Prosocial Behavior
Post by Amanda Engstrom
The takeaway
Orexin neurons in the anterior cingulate cortex generate theta oscillations that transform empathic perception into prosocial behavior, revealing a precise circuit linking emotional understanding to helpful behavior.
What's the science?
Empathy allows animals to perceive and share others’ emotional states and drives prosocial behaviors, which are essential for societal cohesion and well-being. In mice, empathy has been associated with changes in theta oscillations (slow, repeating electrical patterns that coordinate communication between brain regions) in the anterior cingulate cortex (ACC). The ACC has been suggested to be a “central hub” for empathy and prosocial behaviors, projecting into multiple brain regions involved in these complex behaviors.
The neuropeptide orexin regulates arousal, stress, and emotional processing and promotes theta oscillations. However, it remains unclear what upstream circuits modulate these oscillations and how they influence empathy and prosocial behaviors. This week in Science, Kim and colleagues examine how orexin modulates ACC theta oscillations and their relationship to prosocial behaviors.
How did they do it?
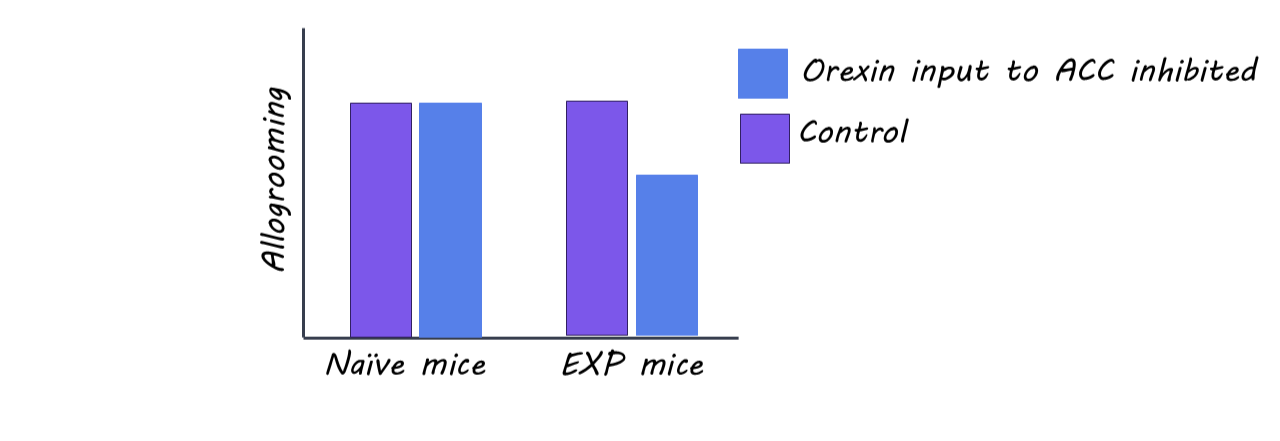
The authors evaluated empathy’s effects on prosocial behavior using an observational fear-conditioning paradigm (one mouse watches another receive a foot shock) combined with a consolation assay measuring allogrooming (the observer mouse grooming the foot-shocked mouse). They tested two paradigms: an experience-dependent observer (EXP), which had previously received a foot shock, and a naïve observer with no prior fear experience. They measured vicarious freezing (the observer mouse freezes at the tone when the experimental mouse receives the foot shock) and allogrooming in both groups and evaluated the impact of consolation on the shocked mouse using an open-field test. To dissect the neuronal mechanisms involved in these behaviors, the authors recorded ACC theta oscillations (5–7 Hz) during behavior. They conducted fiber photometry recordings using a genetically encoded orexin sensor (OxLight1), and using optogenetics, they inhibited ACC-projecting orexigenic circuits in observing mice only during the observation period when the experimental mouse received a foot shock.
What did they find?
Both naïve and EXP observers exhibited vicarious freezing during observation and increased allogrooming after foot shock reunion. However, EXP mice displayed stronger vicarious freezing and more allogrooming compared to naïve mice. Self-grooming increased after observing the foot shock, but did not differ between groups. Notably, emphatic-like behaviors required visual attention - an observer mouse looking away during the foot shock showed no effects. Additionally, the authors found that the increased allogrooming by EXP observers resulted in less anxiety-like behavior in the mice that received the foot shock. Together, these data suggest that shared experiences enhance prosocial behaviors but don’t alter self-directed care.
To dissect the mechanism behind these behaviors, the authors show that both naïve and EXP observers have increased 5- to 7- Hz theta oscillations in the ACC while mice are observing the foot shock and during allogrooming (i.e. empathy and prosocial behavior), but not self-grooming. Concordantly, there was a selective increase in orexin activity at the same time, but only in EXP mice, suggesting that orexin-dependent increases require shared experience rather than observation alone. Inhibiting orexin input to the ACC specifically during the observation period reduced theta power and allogrooming in EXP mice and had no effect in naïve mice. These findings suggest that orexinergic inputs drive ACC theta oscillations to modulate affective empathy and prosocial action when there is shared experience.
What's the impact?
This study found that orexin-dependent increases in ACC theta oscillations link affective empathy to prosocial comforting behaviors. It demonstrates that social behavior relies on specific neuromodulatory rhythms to translate emotional experience into action. Elucidation of this upstream mechanism can provide potential targets for treating disorders that lead to empathy deficits.