The Impact of Corticosteroid Treatments on Hippocampal Function
Post by Baldomero B. Ramirez Cantu
The takeaway
This study provides evidence that corticosteroid treatments can disrupt the circadian regulation of the hippocampus, leading to impairments in hippocampal function and plasticity.
What's the science?
Circadian rhythms are natural 24-hour cycles that regulate various physiological, biological, and behavioral processes in organisms. Corticosteroid treatments are commonly used to manage inflammatory and immunologic disorders, but they can produce side effects such as mental health issues and memory deficits. Despite their widespread use, the underlying mechanisms behind these side effects remain poorly understood. This week in PNAS, Birnie, Claydon and colleagues identify a molecular basis for the memory deficits in patients treated with corticosteroids and provide insights into the influence of corticosteroid treatment on hippocampal function.
How did they do it?
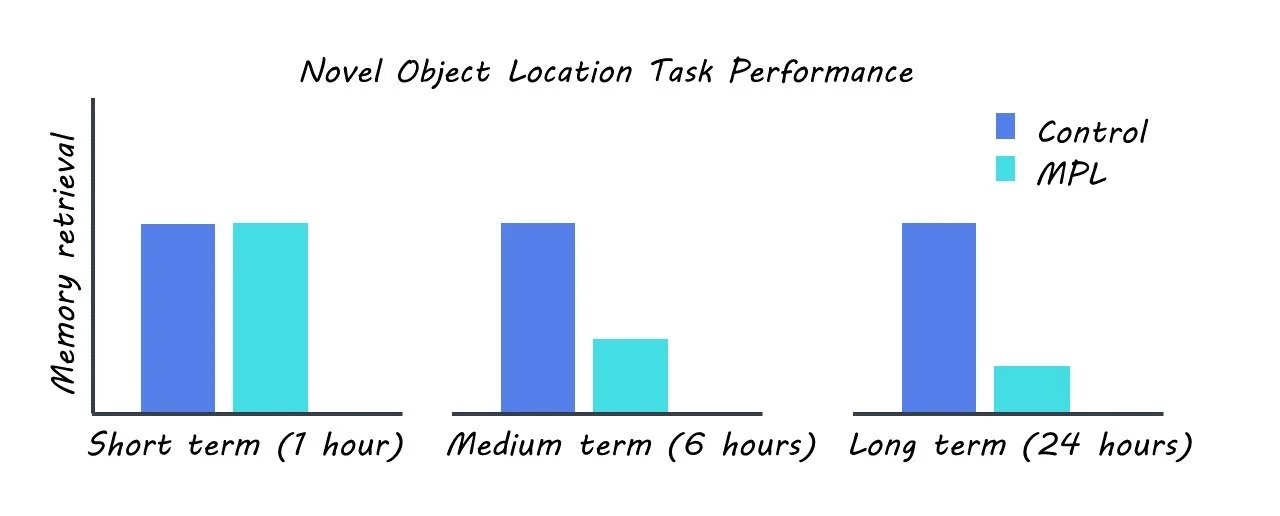
The authors used a rat model to mimic corticosteroid treatment by administration of the commonly prescribed corticosteroid methylprednisolone (MPL). The rats were housed in a 12-hour light-dark and were administered MPL orally. The authors screened for behavioral, genetic, and neurophysiological changes in the MPL group relative to a control group. They monitored changes in locomotion and body temperature between the two groups in order to screen for any potential impacts of the treatment on these behaviors. They also performed a novel object location task to assess changes in short, intermediate, and long-term memory. RNA sequencing was used to measure changes in gene expression in the two groups. Protein levels were assayed using Western blotting. Intracellular ex-vivo recordings were used to explore changes in synaptic plasticity as a result of corticosteroid treatment.
What did they find?
The authors observed that corticosterone treatment caused disruptions in synaptic physiology and gene expression in the hippocampus. Specifically, the authors found that corticosterone treatment impaired long-term potentiation (a measure of synaptic plasticity that is associated with long-term memory), altered the expression of genes involved in circadian regulation and synaptic plasticity, and disrupted the amplitude and frequency of miniature excitatory postsynaptic currents (mEPSCs) in the hippocampus. The authors also showed that corticosterone treatment inhibited NMDAR-dependent plasticity, which is a key mechanism underlying learning and memory in the hippocampus.
In addition, their behavioral task found that rats treated with corticosterone exhibited impaired intermediate and long-term memory, but not short-term memory. The authors further investigated the molecular basis for these effects by analyzing gene expression in the hippocampus. They found that corticosterone treatment dysregulated genes that are known to be crucial for memory processing in the hippocampus and circadian regulation, including CAMKII and CLOCK. Crucially, the authors found that the time of day was a potent modulator of hippocampal activity and that this temporal regulation is disrupted by corticosteroid treatment.
What's the impact?
This study sheds light on the underlying mechanisms of the cognitive side effects of long-term corticosteroid treatments, which are very commonly used to treat many inflammatory and immunologic disorders. Furthermore, these findings could lead to the development of new therapeutic approaches for patients who experience cognitive side effects of corticosteroid treatments.