What is Neurofeedback and How is it Used?
Post by Ewina Pun
Sensory feedback and neuroplasticity
Have you ever tried to walk in a straight line with your eyes closed, or eat a meal in complete darkness? Movement control becomes significantly harder when sensory feedback is limited. When we’re learning, we rely heavily on various forms of feedback, such as tactile, visual, and audio feedback. For some individuals, sensory feedback is lost due to injuries and neural deficits. Fortunately, our brain has the ability to quickly adapt to new circumstances - also known as neuroplasticity.
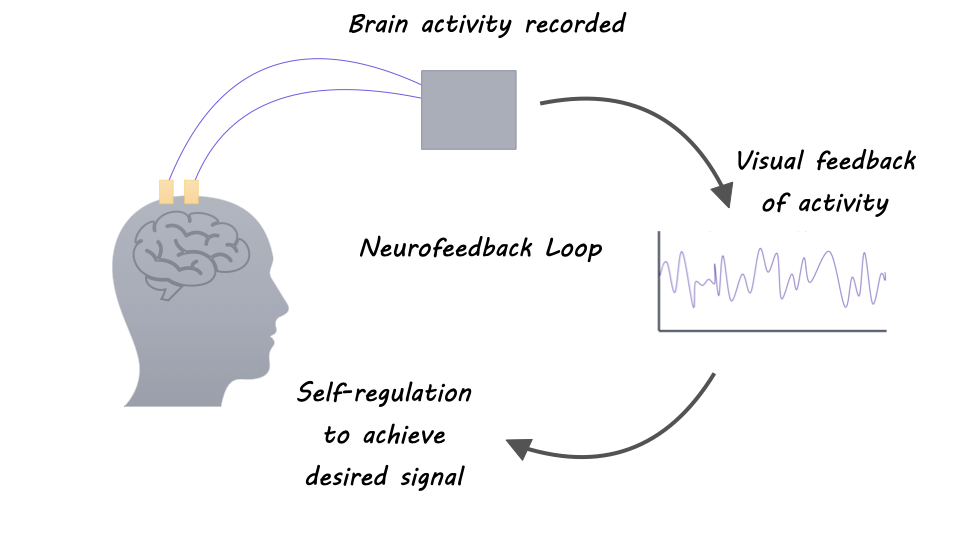
Some researchers study neuroplasticity with neurofeedback: a form of biofeedback that provides a representation of the recorded neural activity as a visual, auditory, or other signal back to the individual in real-time to facilitate self-regulation. In other words, one can modulate their brain activity by integrating information about one’s own brain activity to elicit a different behavior or pathology. Neuroplasticity not only enables cognitive and perceptual learning but also forms the basis of clinical neurorehabilitation. And researchers have begun to investigate the use of neurofeedback for treatments of brain and behavioral disorders.
Neurofeedback to study brain networks
Research suggests that there is a specific network of brain regions involved in self-regulation. Changes in brain activity during neurofeedback can be seen via changes in electroencephalography (EEG) amplitudes and blood oxygen level-dependent signals in functional magnetic resonance imaging (fMRI) in brain areas such as the anterior insular cortex, anterior cingulate cortex, dorsal lateral prefrontal cortex, inferior parietal lobule, basal ganglia, and thalamus. This indicates that neurofeedback involves networks involved in reward processing, cognitive control, and learning and memory.
Neurofeedback for clinical application
Neurofeedback has been extensively studied to treat attention deficit hyperactivity disorder (ADHD). For instance, neurofeedback therapy can provide information about the patient’s brain state to allow the patient to consciously match to and maintain the desired brain state through reinforcement. EEG-based Neurofeedback helps reduce the elevated low-frequency (theta/delta) synchronization observed in children with ADHD and improved ADHD symptoms.
Studies have also found several benefits of neurofeedback for stroke recovery. One study showed that severely impaired, chronic stroke patients learned to upregulate ipsilesional sensorimotor rhythm (SMR) through controlling a brain-computer-interface (BCI) with a hand orthosis, and the neurofeedback training also improved their upper limb functions. In addition, virtual reality in combination with EEG-BCI can offer a more immersive representation of neurofeedback for stroke rehabilitation and increase the perceived embodiment (sense of control as their own). However, more robust evidence is needed to support the efficacy of neurofeedback therapies for ADHD and stroke rehabilitation.
Neurofeedback for BCI motor control
The use of neurofeedback is not exclusive to self-regulate one’s neural state. It is also widely used in BCI for closed-loop motor control. In motor BCI, individuals can directly manipulate an external device with their neural signals being translated into action commands. For example, after training, a BCI maps neural activity patterns to control commands in real-time and provides visual feedback of the current position of the external device being controlled. Such real-time feedback allows users to reevaluate, refine and correct their control. With trial-and-error, people with paralysis can decide which set of mental motor imagery (e.g., imagining controlling a joystick or a computer mouse) was most effective for control. With minimal practice using an intracortical BCI, people with paralysis were able to coordinate movements of a seven degrees-of-freedom robotic arm, control a computer cursor, or functionally stimulate muscles for movement restoration.
Bidirectional closed-loop neurofeedback
In addition to providing just visual feedback, researchers try to directly restore tactile and cutaneous sensations in arm and hand function, which is important during grasping or manipulation of objects. A bidirectional BCI for motor control refers to a system that (1) decodes neural signals in the motor cortex into commands to control a device and (2) provides somatosensory feedback by delivering electric stimulation patterns to the primary somatosensory cortex (S1) or the spinal cord. Microstimulation of the cortical surface of M1 and S1 using high-density electrocorticography (ECoG) has provided tactile sensations such as “buzzing”, “tingling”, “brushing”, “light tapping,” or a “feeling of movement” to participants with paralysis. An adaptive deep brain stimulation framework targeted at the thalamus is capable of concurrent biomimicry stimulation and sensing for better closed-loop therapies for psychiatric disorders, epilepsy, or chronic pain. More research is needed to quantify perceptual qualities and improve naturalistic sensations to provide functional benefits for BCI control.
What’s next?
Neurofeedback is a novel and valuable way to study brain function and neuroplasticity. Further, neurofeedback has exciting potential as a therapeutic tool. Although researchers have begun to understand some of the mechanisms underlying neurofeedback, future research will likely further clarify the psychological and neural underpinnings of self-regulation, which will help to design more-effective neurofeedback technologies for treating a variety of diseases and conditions.
References +
Sitaram et al. Closed-loop brain training: the science of neurofeedback. Nature. (2016)
Saha et al. Progress in Brain Computer Interface: Challenges and Opportunities. Front. Syst. Neurosci. (2021)
Ramos-Murguialday, et al. Brain–machine interface in chronic stroke rehabilitation: a controlled study. Ann. Neurol. (2013).
Zotev et al. Self-regulation of human brain activity using simultaneous real-time fMRI and EEG neurofeedback. Neuroimage (2014).
Collinger et al. High-performance neuroprosthetic control by an individual with tetraplegia Lancet (2013).
Hochberg et al. Neuronal ensemble control of prosthetic devices by a human with tetraplegia. Nature (2006).
Hughes et al. Bidirectional brain-computer interfaces. Handbook of Clinical Neurology (2020).
Ansó et al, Concurrent stimulation and sensing in bi-directional brain interfaces: a multi-site translational experience. Journal of Neural Engineering (2022).