Predicting and Tracking Hallucinations
Post by Leanna Kalinowski
The takeaway
Hallucinations, a common symptom in disorders like schizophrenia, have traditionally been difficult to study given that they cannot be directly observed. Scientists have successfully applied a computational framework to screen for and track hallucinations.
What's the science?
Hallucinations, which are perceptions that occur in the absence of a stimulus, are a hallmark sign of several psychosis-spectrum disorders, such as schizophrenia. Traditionally, hallucinations have been difficult to study given that scientists cannot directly observe them. However, the rise of the field of computational psychiatry field now allows scientists to use mathematical frameworks to better understand the neurological underpinnings of psychosis-spectrum disorders.
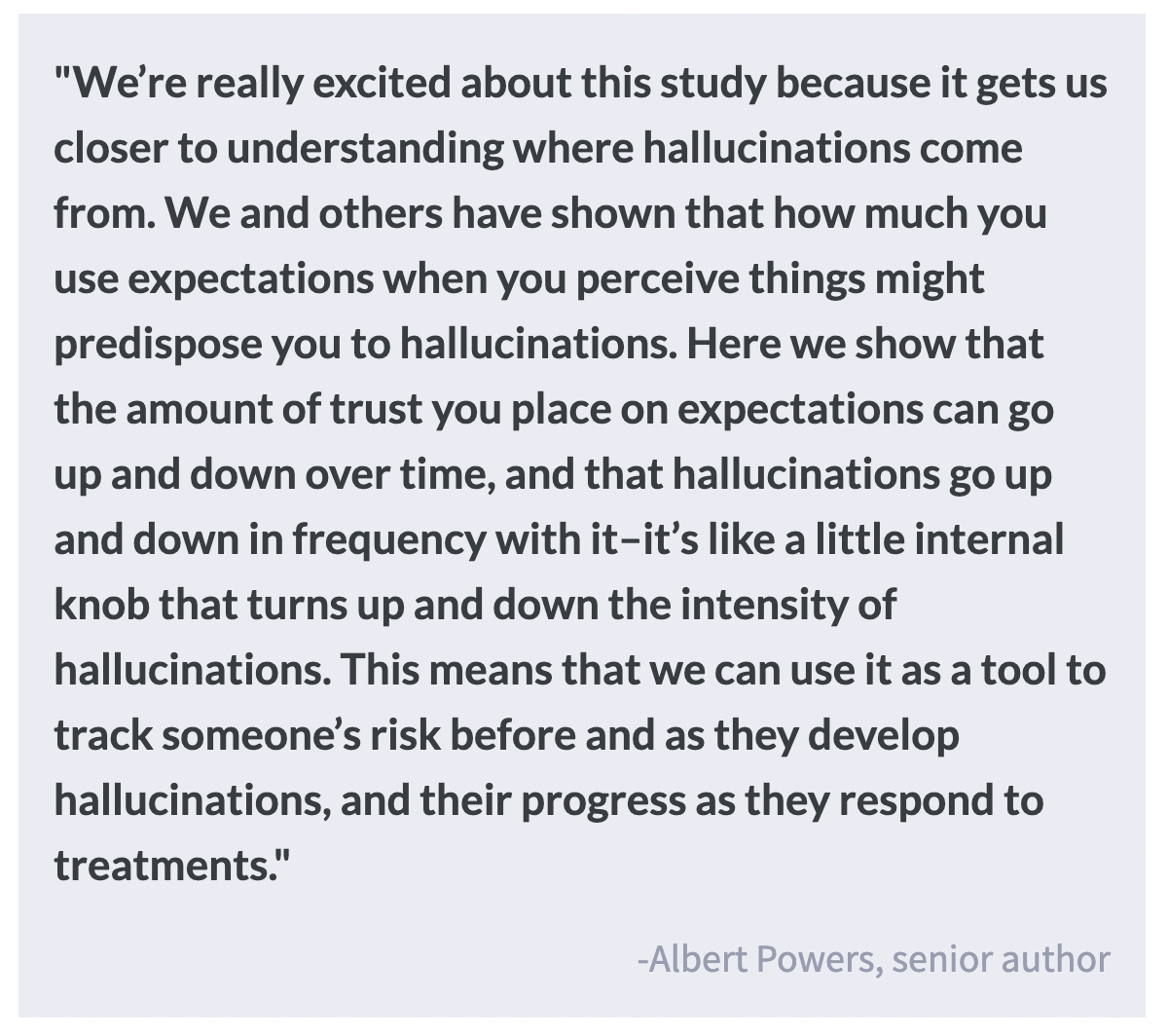
One such framework, predictive processing theory, shows promise as a tool for better understanding hallucinations. In this framework, “perception” is described as the process of determining the cause of one’s sensations by considering (1) one’s internal expectations of their surroundings based on prior knowledge (called “priors”) and (2) the available sensory evidence that is weighted by the participant’s certainty in the source of the information. Evidence suggests that hallucinations arise when the priors are over-weighted compared to incoming sensory evidence, but this exact relationship is unclear. This week in Biological Psychiatry, Kafadar, Fisher, and colleagues used mathematical modeling to determine the relationship between these over-weighted priors and susceptibility to hallucinations.
How did they do it?
First, 458 participants were screened for the presence of auditory hallucinations and separated into two groups: hallucinators and non-hallucinators. Then, they completed the Auditory Conditioned Hallucinations task, where participants are first trained to associate a visual pattern with an auditory tone. Once the association is learned, the researchers then recorded the conditioned hallucination rate, which is the proportion of times that the participants reported hearing the tone when the visual pattern was displayed, without the tone. Finally, a subset of the hallucinators group was invited back to the lab 6-12 months later to determine whether performance on this task is related to changes in symptom severity.
What did they find?
The researchers found that conditioned hallucination rates were a predictor of the frequency of self-reported hallucinations. These rates were sensitive to hallucination state and the over-weighting of priors compared to incoming sensory evidence. They also found that conditioned hallucination rates and prior weighting are higher in the hallucinator group. Changes in these rates were further associated with changes in the frequency of reported hallucinations at the follow-up test, suggesting that this approach may inform future clinical screening tools.
What's the impact?
Taken together, these results indicate that conditioned hallucination rates and over-weighting of priors can be used as markers of hallucination status. This can be useful when tracking the development, trajectory, and treatment response of psychosis-spectrum disorders.