How Deep Brain Stimulation Affects Decision-Making in Parkinson’s disease
What's the science?
How much time should we give ourselves to make a decision? For example, when faced with a difficult decision, we might give ourselves more time to garner more evidence before we reach the ‘decision threshold’ and decide. One brain region involved in adjusting our decision threshold (meaning we take more or less time before the decision) is the subthalamic nucleus (STN). Deep brain stimulation (DBS) of the STN is often performed to reduce motor symptoms in Parkinson’s disease, however, a negative side effect can be impairment in adjusting the decision threshold, leading to impulsive responses. This week in Current Biology, Herz and colleagues conducted a study in patients with DBS electrodes for Parkinson’s placed in the STN, in order to assess how stimulation of this site affects the decision threshold.
How did they do it?
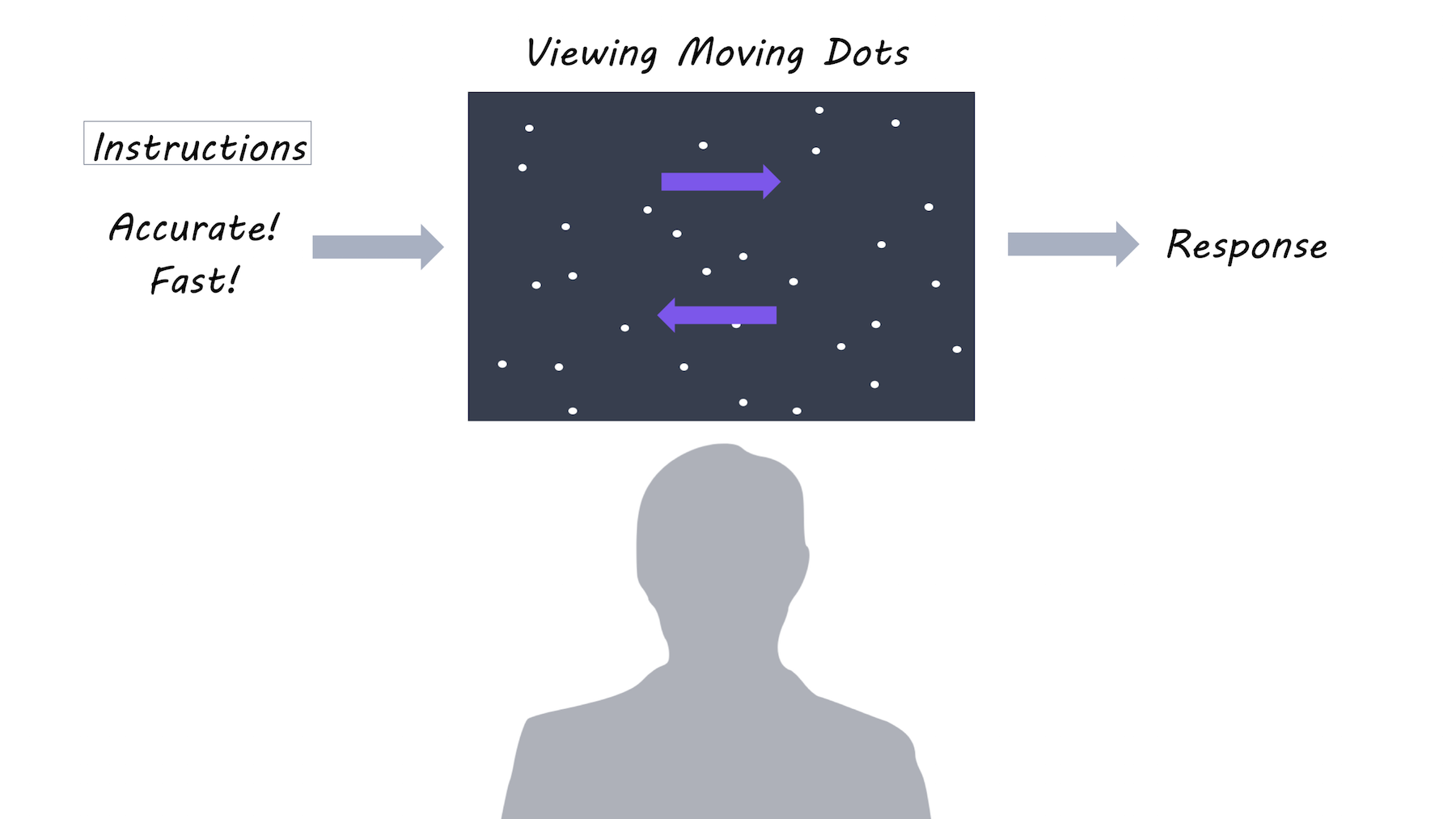
Ten patients with Parkinson’s participated in the study after undergoing surgery to implant electrodes for DBS in the STN. Each patient performed a decision-making task: 1) when DBS was off 2) with DBS on continuously and 3) with ‘adaptive DBS’ where DBS only turns on when necessary. The decision-making task involved looking at dots moving on a screen, and deciding whether the majority of dots were moving to the left or to the right. There were two task conditions and two forms of instruction: In the easy condition, 50% of the dots moved in the same direction, while in the difficult condition only 8% of dots moved in the same direction. Participants were also instructed to focus on either speed or accuracy of their decision.
What did they find?
When DBS was off, participants responded more slowly during the difficult task and when instructed to focus on accuracy (versus speed). However, when DBS was on, slowing during a difficult task was diminished, but slowing due to focus on accuracy remained the same. During adaptive DBS, stimulation came on at different times across trials (when beta activity happened to be high). When the DBS stimulation came on during a 400-500 ms time window after the moving dots appeared on the screen, the time required to make a decision (usually increased during the difficult task) was most diminished, suggesting that the effect of stimulation is confined to a short time window. Using ‘drift diffusion modelling’, they found that stimulation affected the decision threshold time specifically, as opposed to, for example, the motor response time. While DBS was off, beta activity increased after presentation of the dots during the difficult condition, and was related to the decision threshold, but these effects were abolished during stimulation. This indicates that DBS may be lowering the decision threshold by changing the relationship between STN activity and threshold adjustments.
What's the impact?
These results are the first to show that the STN may be directly involved in decision thresholds (how much evidence we need before we reach a decision). During a narrow time window, the STN adjusts decision thresholds based on the anticipated difficulty of the decision. This may be a mechanism by which decision-making is impaired in people with Parkinson’s who have DBS.
P. Brown et al., Mechanisms Underlying Decision-Making as Revealed by Deep-Brain Stimulation in Patients with Parkinson’s Disease. Current Biology (2018). Access the original scientific publication here.