Inflammation Triggers Somatic Bassoon Protein Accumulation in Multiple Sclerosis
Post by Shireen Parimoo
What's the science?
Multiple sclerosis (MS) is a neuroimmunological disease characterized by myelin (insulation for neuronal axons) damage due to inflammation in the central nervous system. This damage impairs neuron function and can cause neuronal death, resulting in symptoms like fatigue, loss of coordination, and cognitive impairment. The molecular mechanism by which inflammation results in neuronal loss is not clear because isolating damaged neurons from neuronal tissue has been a methodological challenge, making it difficult to determine the involvement of various proteins in this process. This week in Nature Neuroscience, Schattling and colleagues used neuron-specific profiling of messenger RNA to investigate the gene expression profiles of different cell types in inflamed tissue from MS patients and model organisms with MS.
How did they do it?
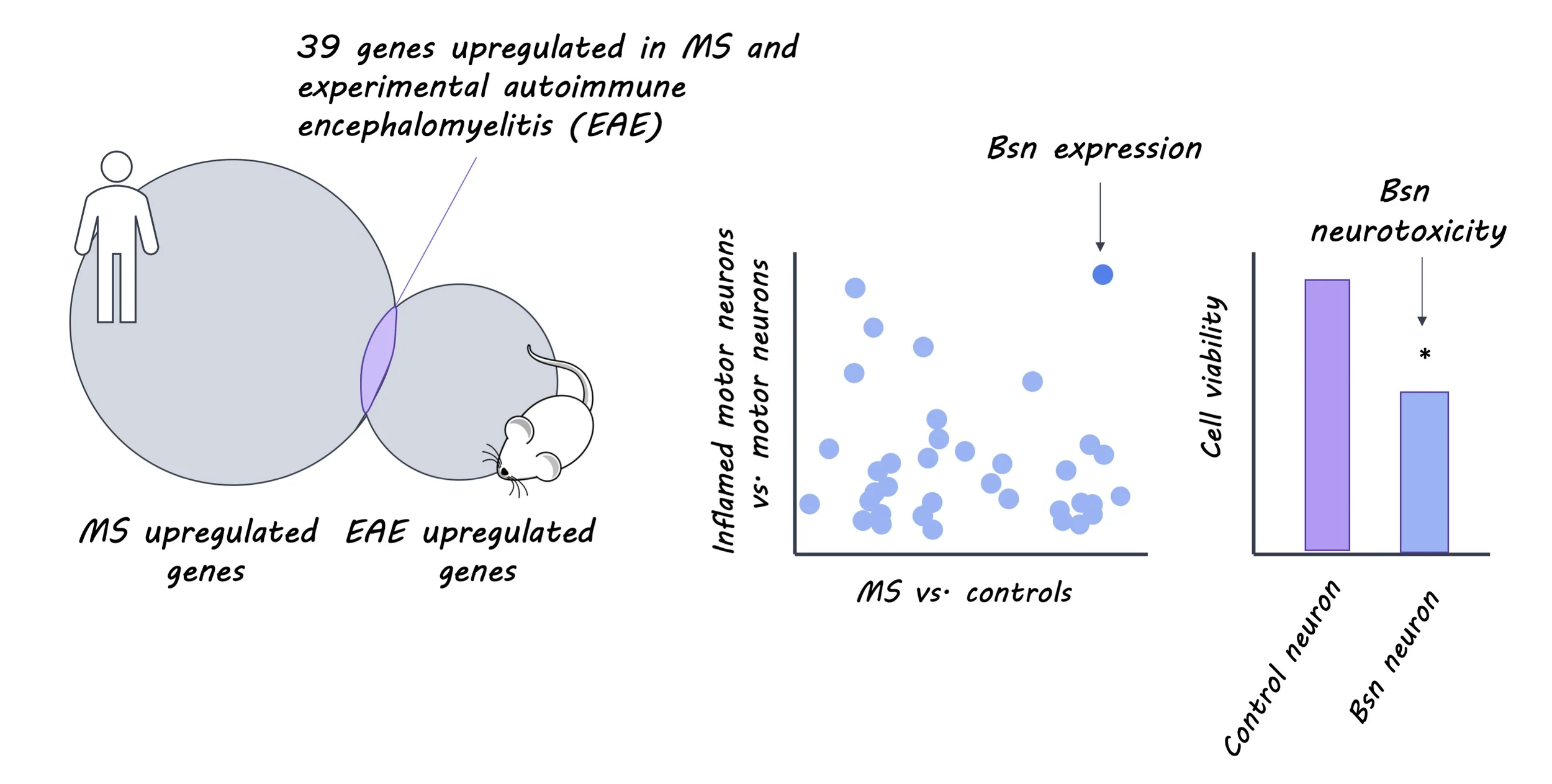
The authors first examined the genes expressed by neurons in inflamed spinal cord tissue. Mouse models of MS were created by inducing experimental autoimmune encephalomyelitis (EAE), which triggers inflammation and the demyelination of neurons, and results in partial or full paralysis. Translating ribosome affinity purification was used to isolate messenger RNA of different cell types in normal and inflamed cervical spinal tissue and gene set enrichment analysis was performed to determine whether certain genes were over- or under-expressed in those cell types. To identify which genes were up- and down-regulated in humans, they used a microarray dataset of MS patients to compare genes expressed in chronic MS plaques and in normal tissue. They then compared the overlap in genes expressed in both humans and mice using homology mapping, which is a method used to identify the genes shared by two different species. The Bassoon (Bsn) protein – a pre-synaptic scaffolding protein found in synaptic active zones – was highly expressed in inflamed motor neurons. The authors used immunohistochemistry to examine Bsn expression and localization in the mouse and human spinal cord. They further explored the cellular and behavioral effects of high and low Bsn expression and Bsn deletion (knockout) in mice and transgenic fruit flies. Cellular measures included energy metabolism, Bsn accumulation, and cellular survival and number. Behavioral measures included EAE symptomatic recovery in mice and climbing ability in flies. Finally, they investigated whether removing Bsn from neurons would rescue the adverse effects of inflammation and enhance neuronal survival by administering the ubiquitin carboxyl-terminal hydrolase 14 inhibitor (IU1), which allows proteins like Bsn to be degraded.
What did they find?
The authors identified 354 up-regulated and 448 down-regulated candidate genes in motor neurons found in inflamed mouse spinal cord tissue. The majority of the up-regulated genes were those involved in protein breakdown pathways, while the down-regulated genes were primarily those involved in energy metabolism, suggesting that the cellular energy metabolism process is impaired during inflammation. In humans, 11% of the up-regulated genes identified in chronic MS plaques overlapped with those observed in mice, including Bsn. There was a greater concentration of Bsn in spinal cord tissue obtained from EAE mice and MS patients than in wild-type mice and control participants. Interestingly, in these groups Bsn accumulated in the cell bodies of the inflamed motor neurons, even though it is typically located in the active zone of presynaptic neurons.
Increased Bsn expression in mouse cells was associated with Bsn accumulation in cell bodies, reduced energy metabolism, and reduced cellular survival. Similar to humans and mice, Bsn accumulation was observed in the neuronal cell bodies of transgenic flies, along with reduced climbing ability and increased mortality. On the other hand, mice with Bsn deletion (knockout mice) showed faster and better recovery from EAE symptoms compared to wild-type EAE mice. The knockout mice also had more axons and neurons in the spinal cord and fewer injured axons, but the same number of immune cells as wild-type mice. This suggests that the immune response to inflammation is not affected by knocking out the Bsn protein, and that Bsn likely plays a crucial role in determining neuronal fate. Consistent with this, administering IU1 facilitated Bsn degradation in neuronal cell bodies of mice and even restored pre-synaptic Bsn localization. Furthermore, mice that were administered IU1 had a larger number of axons and neurons, as well as a faster and better recovery trajectory than mice that were given a vehicle solution. These findings indicate that Bsn accumulation in the cell body might exacerbate the effects of inflammation by interfering with cellular energy metabolism and other intracellular processes.
What's the impact?
This study is the first to demonstrate that inflammation triggers Bassoon accumulation in neuronal cell bodies of both MS patients and model organisms. Importantly, both the cellular and symptomatic effects of inflammation can be partially reversed by promoting Bsn degradation. These findings have important implications for our understanding of the mechanisms underlying MS as well as potential targeted treatments for mitigating the symptoms of the disease.
Schattling et al. Bassoon proteinopathy drives neurodegeneration in multiple sclerosis. Nature Neuroscience (2019). Access the original scientific publication here.