The Impact of Deep Brain Stimulation on Brain Networks and Treatment Outcomes in Treatment-Resistant Depression
Post by Kulpreet Cheema
The takeaway
Deep brain stimulation of the subcallosal cingulate gyrus in patients with depression induces changes in brain network properties, which is correlated with improvements in depression symptoms.
What's the science?
Treatment-resistant depression (TRD) is a severe form of depression that does not respond to standard treatments. Deep brain stimulation (DBS) is an emerging therapy that involves implanting electrodes in specific brain regions to modulate neural activity and potentially alleviate depressive symptoms. The effectiveness of DBS varies among patients, and personal factors might be the key to understanding this variability. This week in Molecular Psychiatry, Ghaderi and colleagues aimed to understand how DBS affects brain networks in TRD patients using electroencephalography (EEG), a non-invasive technique that measures the brain’s electrical activity.
How did they do it?
The study involved twelve TRD patients who underwent subcallosal cingulate gyrus (SCG) DBS. The researchers collected resting-state EEG data and Hamilton Depression rating scale data from participants at three sessions: before surgery, one to three months after surgery, and six months after surgery. During the second and third sessions, EEG data was collected with DBS turned on and off sequentially.
To analyze brain network properties, the researchers used an analytical method known as graph theoretical analysis to decipher whether there were distinct differences in brain network properties between patients who responded positively to the DBS treatment (responders) and those who did not (non-responders). Using EEG to study oscillatory activity in brain networks, the authors analyzed four different frequency bands — the delta and alpha bands were of particular interest due to their relevance to depression.
What did they find?
The study revealed several important findings. First, the baseline brain network properties in patients — especially in the delta and alpha frequency bands — were associated with the outcomes of DBS treatment. Responders to DBS showed lower levels of network segregation, integration, and synchronization at baseline compared to non-responders. Second, DBS led to changes in brain network properties over time, characterized by increased integration and synchronization of brain regions. These changes were particularly evident in the delta frequency band.
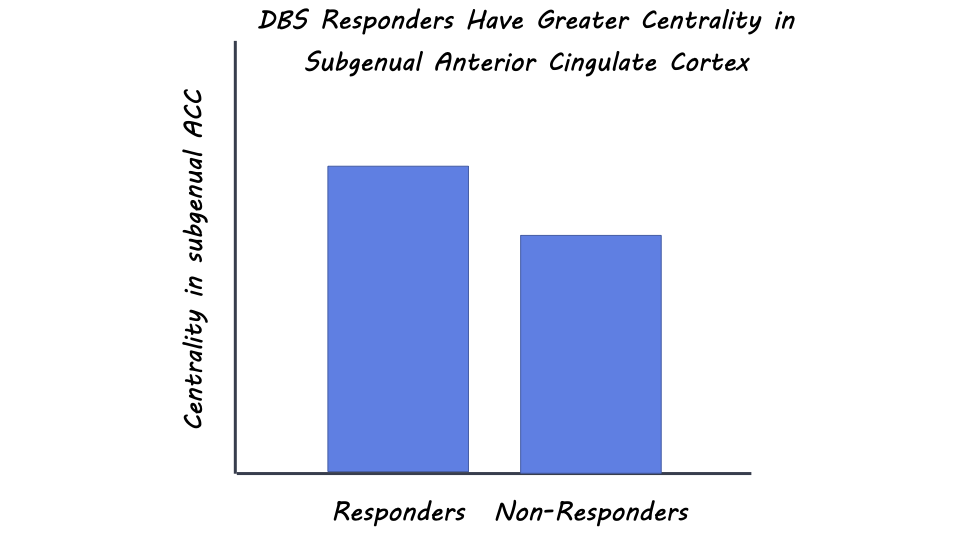
Third, the researchers found that responders had higher centrality - a measure of the importance of a node in a network for information propagation - in the subgenual anterior cingulate cortex (ACC), a brain region associated with depression symptoms. DBS led to a reduction in centrality in this region, which correlated with treatment response. Other brain regions, like the primary somatosensory cortex and parahippocampal gyrus, also showed alterations in centrality, indicating their involvement in TRD and DBS response.
What's the impact?
This study sheds light on the complex effects of DBS on brain networks in treatment-resistant depression. By using EEG and advanced analytical techniques, researchers identified specific brain network features associated with treatment response and demonstrated how DBS induces changes in these networks over time. The findings provide valuable insights into the potential mechanisms underlying the therapeutic effects of DBS in TRD patients. This knowledge could contribute to the development of personalized treatments for individuals with treatment-resistant depression, ultimately offering new hope for improved mental health outcomes.