In Vivo Imaging of REM Sleep Behavior Disorder
What's the science?
Rapid Eye Movement (REM) Sleep Behavior disorder often precedes the development of Parkinson’s disease, which is characterized by the accumulation of the protein alpha-synuclein inside of neurons. Recent research suggests that alpha-synuclein first accumulates in peripheral autonomic neurons (in the gut) and in the olfactory bulb (in the brain). Alpha-synuclein is hypothesized to then spread to the brain via autonomic nerve fibres, causing further damage via neuron-to-neuron transmission from peripheral neurons through the spinal cord and brainstem to the brain. This week in The Lancet Neurology, Knudsen and colleagues imaged pathology in the periphery and in the brain of multiple affected systems in patients with REM Sleep Behavior Disorder to better understand how alpha-synuclein might progress prior to the onset of the classical motor symptoms in Parkinson’s disease.
How did they do it?
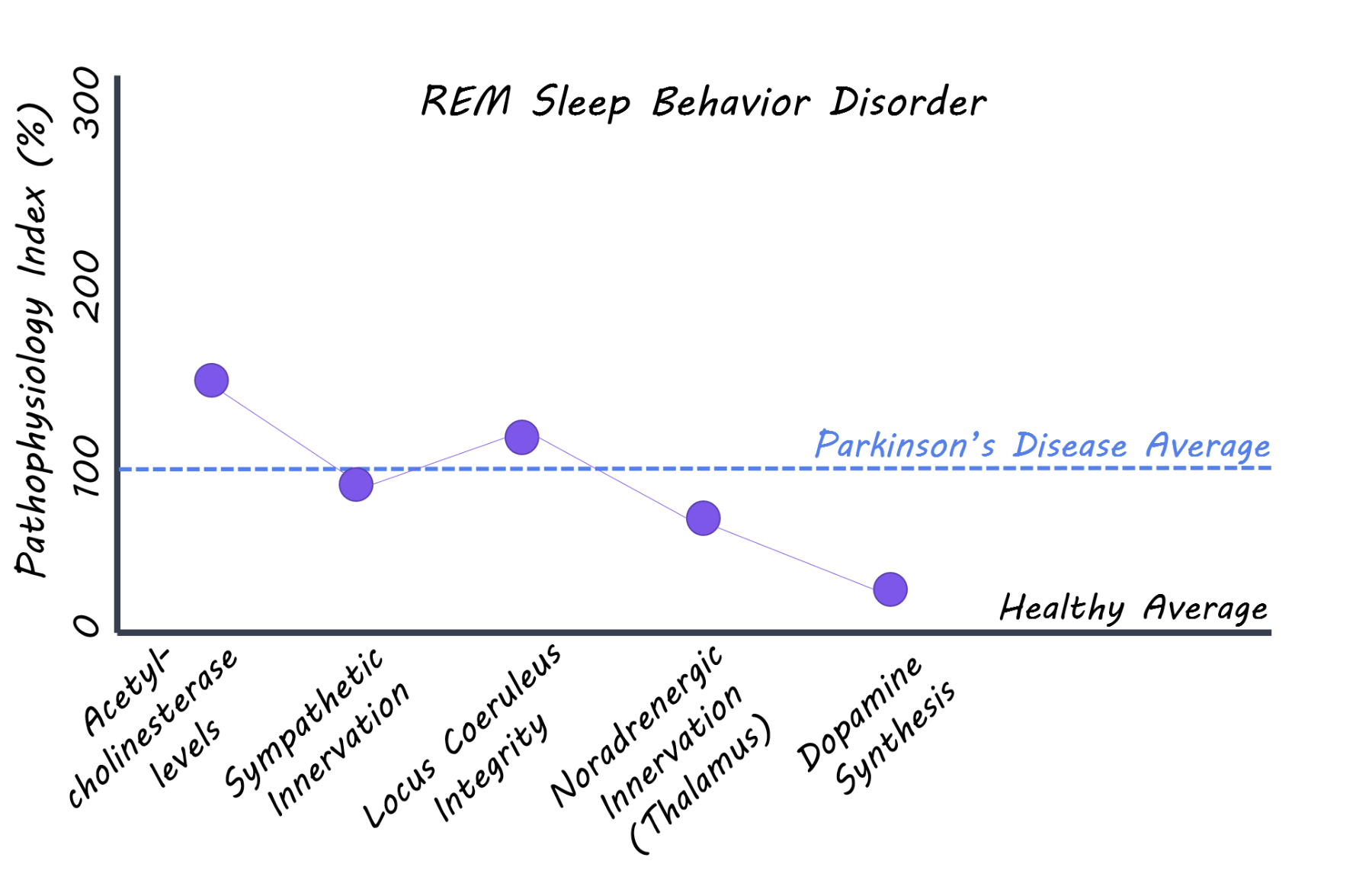
Patients with REM sleep behavior disorder (with no signs of parkinsonism or dementia), patients with diagnosed Parkinson’s disease, and a healthy control group all aged 50-85 years were recruited for the study and underwent a series of imaging scans. 11C donezepil (a radiotracer that measures acetylcholinesterase concentrations) PET imaging was used to assess the level of cholinergic innervation in the gut reflecting, in part, parasympathetic nerve fibres. 123I-MIBG (radiotracer measuring cardiac innervation) scintigraphy was used to measure the level of sympathetic innervation of the heart. Neuromelanin sensitive MRI was used to evaluate the integrity of neurons in the locus coeruleus (brainstem nucleus). 11C-methylreboxetine (a radiotracer that binds to noradrenaline transporters) was used to measure the level of noradrenergic nerve terminals in the locus coeruleus projections to the thalamus. Lastly, 18F-DOPA PET (a radiotracer that measures dopamine synthesis) was used as a measure of dopamine neuron integrity in the brain. Imaging measures between REM sleep behavior disorder and control groups were compared. Imaging multiple systems — from the peripheral autonomic nervous system through to the brainstem — allowed for an assessment of the degree of damage to these important neuronal systems.
What did they find?
Patients with REM sleep behavior disorder had significantly less cholinergic innervation in the small intestine and the colon compared to the healthy control group, while there was no significant difference between REM sleep behavior disorder and Parkinson’s disease patients. Patients with REM sleep behavior disorder also showed significantly lower cardiac innervation compared to controls and lower neuron integrity in the locus coeruleus (brainstem), while again no difference was seen when compared to the Parkinson’s disease group. Locus coeruleus neuron innervation of the thalamus was lower in REM sleep behavior patients compared to controls, but no difference was seen in any other comparison. In contrast, the dopamine synthesis capacity measured by 18F-DOPA PET was normal in 70% of REM sleep behavior patients, while all patients with Parkinson's disease show markedly reduced dopaminergic function in the striatum (the cause of their motor symptoms). Combined, these observed differences support the hypothesis that the autonomic nerve fibres of the gut and other internal organs are affected many years prior to the Parkinson's disease diagnosis. The finding also supports that the initial alpha-synuclein damage in Parkinson's disease may originate in the gut and spread via the autonomic nervous system to the spinal cord and brainstem.
What's the impact?
This is the first comprehensive imaging study of patients with REM sleep behavior disorder (prodromal Parkinson’s disease) to assess damage in vivo to the autonomic nervous system and multiple brainstem systems. The study supports the hypothesis that alpha-synuclein pathology may initially form in peripheral organs and spread neuron-to-neuron via the autonomic nervous system to the brainstem. If Parkinson's disease pathology truly originates in nerve terminals of peripheral organs, different strategies of neuro-protection might be possible, including the use of drugs which do not cross the blood-brain barrier or modification of the intestinal microbiome.
Knudsen et al., In-vivo staging of pathology in REM sleep behaviour disorder: a multimodality imaging case-control study. The Lancet Neurology (2018). Access the original scientific publication here.